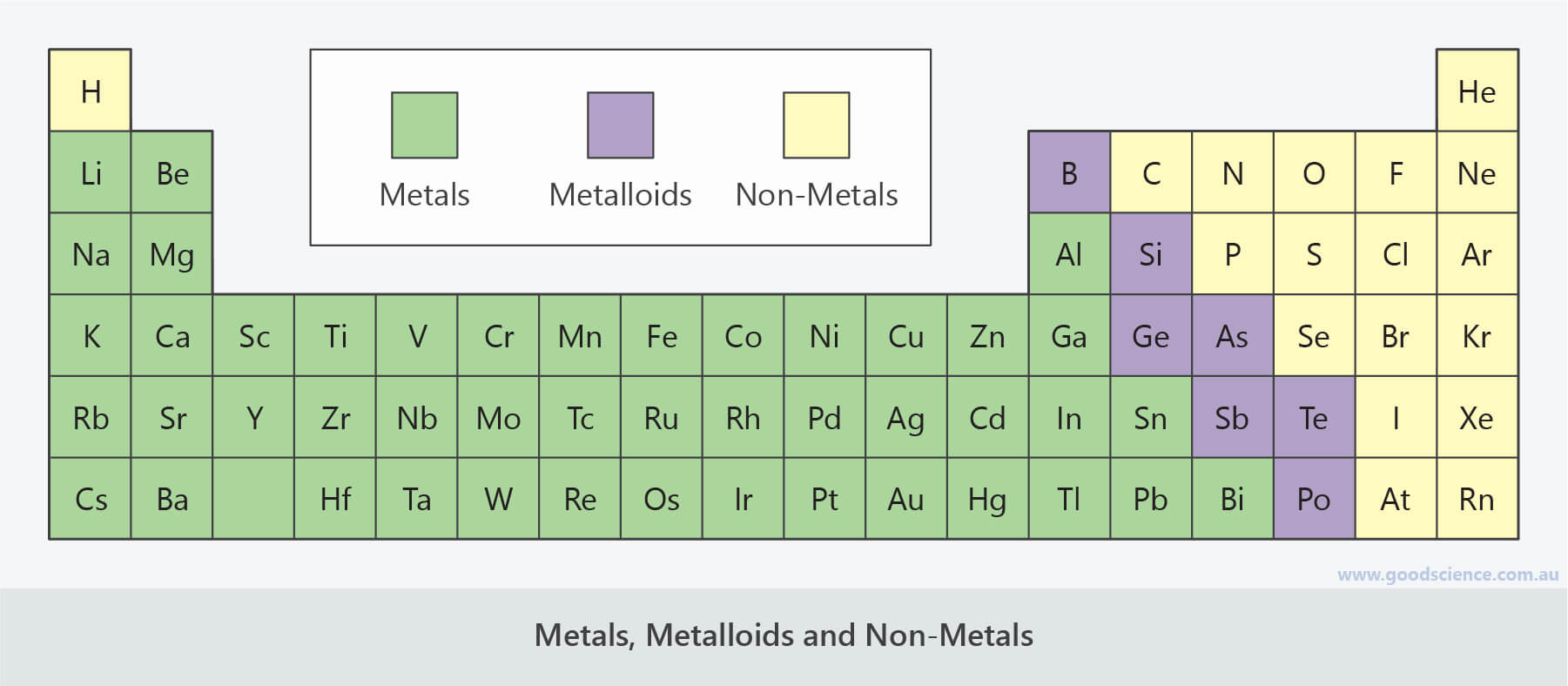 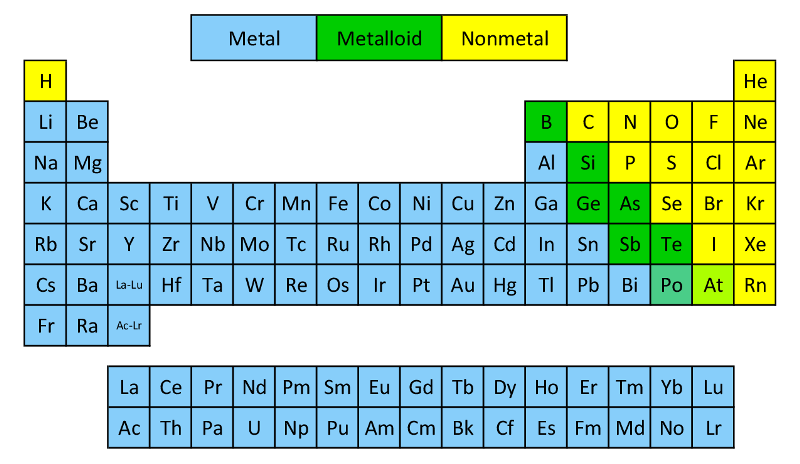
One exception is silicon, which is brittle. Semimetals are typically malleable and ductile.Semimetals/metalloids have high lattice dielectric constants and high diamagnetic susceptibilities.These elements have lower electrical and thermal conductivity than metals.Exceptions are silicon and germanium, which are true semiconductors, as they can conduct electricity under the right conditions. Semimetals tend to make excellent semiconductors, although most of the elements themselves are not technically semiconducting.A band gap separates a filled valence band from an empty conduction band. However, the defining characteristic of metalloids is not so much their position on the periodic table as the extremely small overlap between the bottom of the conduction band and top of the valence band. 
Semimetals or metalloids are found in a zig-zag line on the periodic table, separating the basic metals from the nonmetals. Metalloids tend to be shiny, brittle solids that act as insulators at room temperature but as conductors when heated or combined with other elements.Metalloids are used to make semiconductors, ceramics, polymers, and batteries.Some scientists also consider tennessine and oganesson to be metalloids. Usually, the semimetals or metalloids are listed as boron, silicon, germanium, arsenic, antimony, tellurium, and polonium.On the periodic table, metalloids are found along a zig-zag line between boron and aluminum down to polonium and astatine.Metalloids are chemical elements that display properties of both metals and nonmetals.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
